
As an innovative pharmaceutical industrialization base, Jiangsu Allist mainly undertakes the commercial production of Furmonertinib Mesilate Tablet with an annual production capacity of 100 million tablets at present.
Conduct the production of clinical samples and pilot-scale research in the new drug research and development phase.
Jiangsu Allist has 17,000 square meters of production workshop and laboratory, advanced equipment and facilities, all the key production equipment and testing instruments are international first-class brands.
The design concept of multiple varieties, small batch size and high added value has been applied to antitumor solid dosage form workshop.Various granulation processes include wet granulation, dry granulation, one-step granulation, extrusion-spheronization etc. Two dosage forms: tablet and capsule. Multiple packaging such as Aluminum-Plastic, Aluminum-Aluminum and bottling-packaging.
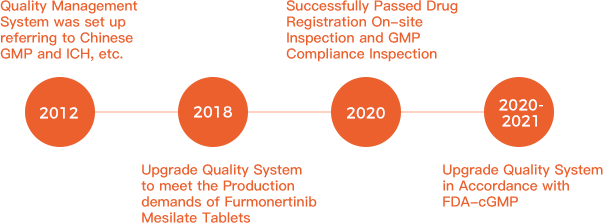
Base on the perfect quality system and operation management, Jiangsu Allist has successfully not only passed the new drug registration on-site inspection and GMP Compliance Inspection but also passed overseas customer audit for many times.
Document System: 19 categories and over 700 documents, including Organization and Personnel, Equipment and Plant Facilities, Qualification and Validation, Production Management, Quality Management, Raw Materials and Drug Product, Computerized System etc.
Self-Inspection: Strictly Self-Inspection management (at least once a year), practical execution plan, actively invite professional institutions and experts for simulation inspection from time to time.
Set up a standardized QC laboratory in accordance with GMP requirements. All key instruments are international first-line brands and under network edition management.
Computerized system validation has been completed for all key instruments.
All analysts have been trained in GMP and analyze operational skills.
Standardized laboratory management and operation.
Pay attention to environmental protection,occupational health and safety in the production process
Use closed feeding special interface
Closed transfer of dedicated PE bags to minimize the contamination and cross contamination in production process

